2024 Better World Project Awards
AUTM's
Better World Project highlights the global impact of research commercialization and the vital role that technology transfer plays in that process. The annual Better World Project Award honors the exemplary work of one technology transfer office from the stories submitted the previous year. More than 50 stories were submitted to the
Better World Project last year. The Better World Project Committee narrowed them down to three very worthy finalists.
The
2024 Better World Project Award winner is:
University of South Florida’s NEWgenerator Provides Safe Sanitation to Remote Locations

Hundreds of elementary school students in South Africa lack access to clean water and sanitation due to their remote locations. Now those students and others have a new option for safe sanitation: NEWgenerator, designed and developed by Daniel Yeh, PhD, and his team at the University of South Florida (USF). The solar-powered innovation provides off-grid sanitation and generates nutrients, energy and water by safely recovering them from human wastewater. Recognizing the potential impact of this breakthrough technology, the USF Tech Transfer Office (TTO) collaborated with the inventors, lawyers and the university to secure IP protection. To ensure this technology could reach the people who needed it most, the TTO team also negotiated non-exclusive license agreements with other countries and partnered with organizations like the Bill & Melinda Gates Foundation.
Meet the finalists:
Inspired by His Son, Boston University Professor Invents Bionic Pancreas to Manage Type 1 Diabetes
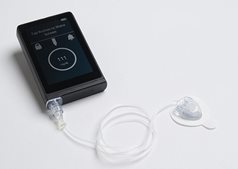 Those with type 1 diabetes have relied on continuous glucose monitors and insulin pumps to help regulate and manage the disease. Yet what if there were a wearable device that, as needed, automatically delivered insulin to a user? That’s what Ed Damiano, PhD, a professor of biomedical engineering at Boston University, has created. In May 2023, the FDA cleared the iLet bionic pancreas that Damiano developed over two decades. The iLet bionic pancreas is an insulin delivery system that works with glucose monitor to reduce the need to make decisions about diabetes management. From his first idea, to FDA approval, Damiano worked in partnership with the university’s tech transfer office, which received its first of many disclosures in 2004.
Those with type 1 diabetes have relied on continuous glucose monitors and insulin pumps to help regulate and manage the disease. Yet what if there were a wearable device that, as needed, automatically delivered insulin to a user? That’s what Ed Damiano, PhD, a professor of biomedical engineering at Boston University, has created. In May 2023, the FDA cleared the iLet bionic pancreas that Damiano developed over two decades. The iLet bionic pancreas is an insulin delivery system that works with glucose monitor to reduce the need to make decisions about diabetes management. From his first idea, to FDA approval, Damiano worked in partnership with the university’s tech transfer office, which received its first of many disclosures in 2004.
Gene Therapy from Nationwide Children’s Hospital is First to Receive FDA Approval for Duchenne Muscular Dystrophy
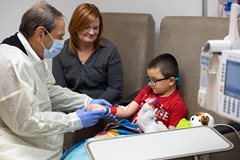
In children with Duchenne muscular dystrophy (DMD), a gene mutation causes progressive weakness and damage to muscles—including muscles responsible for heart function and breathing—that lead to death in early adulthood. Now children with DMD have a new treatment option, thanks to a gene therapy developed at Nationwide Children’s Hospital that is the first DMD treatment to target the genetic cause of the disease in addition to the symptoms. The Office of Technology Commercialization initially entered into an option agreement with Sarepta Therapeutics in late 2016, then licensed the technology in 2018 following promising positive Phase I clinical trial results. This licensing deal helped to move the therapy into clinical trials and further development quickly and efficiently.
If you want to be in the running for the Better World Project Award next year, please visit www.autm.net/BWP to submit your technology transfer office's success story! We'd love to see your story here in 2025.